- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Shree swami samarth songs online
- Watch dogs legion season pass
- Toon boom animate pro 2 effects not rendering
- James patterson books in order murder is forever
- Bully ps2 iso
- Paint tool sai 1-2-5 free
- Presentation assistant alternative
- Lumerical fdtd solutions tutorial
- Warcraft 3 pokemon tower defense english
- Dr farhat hashmi lectures 2015
- Microsoft office 2003 professional edition free download
- Saath nibhana saathiya episodes 2016
- Adobe premiere pro cs5 system requirements
- Sim girl version 7
- Pes 2011 soundtracks download
- Maxis simtown
- 399 ev xps peak
- Pdf xchange viewer review
- Mbox 2 pro craigslist
- Terrarium tv app 1-9-10
- Jacksepticeye simulacra 2
- New Page
- Octopath traveler ost vgmdb
- How to make a dvd from adobe premiere pro cc
- Apex sql doc
- Chalte chalte movie review
- Ggu law career services
- Autocad 2009 windows 7 patch
- Sketchup pro 7 mac
- Autodesk hsmworks professional 2015
- Kingdom rush frontiers achievements
- Visual studio server explorer window
- Magicad autocad
- Blaupunkt amp7502
- Elgato eyetv 250plus
- Visualizer 3d proofing software
- What does cities skylines deluxe edition have
- Etka 7-5 downlad
- Autodesk autocad lt 2014 sp1
- Wic reset utility keygen crack
- Cool robot fonts
- Mototrbo cps 2-0 latest version
- Simcity pc gameplay
- Rusty lake hotel room order
- Gamecube iso tool usb loder gx
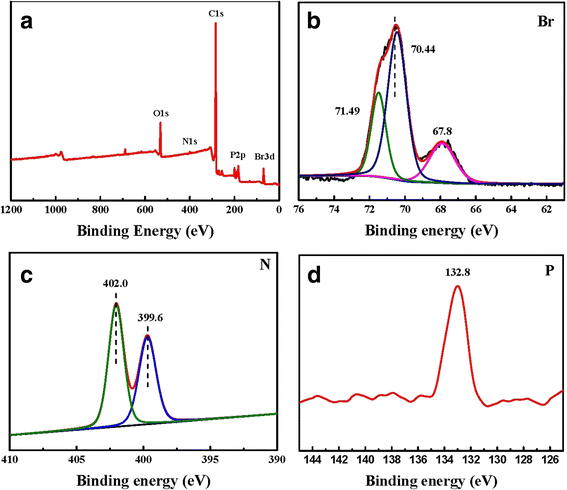
The NPHT and TNDDE SAMs have been shown to behave differently to X-ray exposure. Furthermore, the influence that the phenyl group has on the chemical modification induced by the X-ray irradiation on the SAMs was investigated by comparing the XPS results obtained from irradiation on a NO2-aromatic-terminated SAM (6-(4-nitro-phenoxy)-hexane-1-thiolate (NPHT)) and NO2-aliphatic-terminated SAM (thioacetic acid S-(12-nitrododecyl) ester (TNDDE)).
#399 EV XPS PEAK SERIES#
Also, there was no evidence of the Molecules desorbing from the substrate for any of the SAMs studied during the X-ray irradiation as shown by no change in the S 2p and C Is XPS spectra taken during the X-ray irradiation.Ībstract = "The chemical modification caused by prolonged exposure to X-rays on a series of para-substituted phenyl moieties (-NO2, -CN, -CHO, -COOH, -CO2Me, and -(CO2Bu)-Bu-1) at the surface of thiolate-Au self-assembled monolayers (SAMs) has been investigated by X-ray photoelectron spectroscopy (XPS). The S 2p and O Is spectra remained unchanged during the irradiation suggesting the conversion of the -(CO2Bu)-Bu-1 to the -COOH moiety, although the conversion was not complete because the tertiary butyl moiety contributes 25% to the total carbon content of the SAM. No change was observed on the other functionalized monolayers apart from the -CO(2)(1)Bufunctionalized monolayer, where after 100 min of X-ray irradiation approximately 11% of the carbon content was lost. On the TNDDE SAM, XPS showed the -NO2, photoelectron peak again decreasing with prolonged X-ray irradiation whereas no peak was observed at 399 eV therefore, the -NO2 moieties are selectively cleaved. The irradiation of the NPHT SAM led to the reduction of the nitro (-NO2) moiety to the amine (-NH2) moiety, as shown by the decrease in the intensity of the N Is photoelectron peak for -NO2 (406 eV) in the XPS spectra with the concomitant increase in the N Is photoelectron peak for -NH2 (399 eV). Angular dependent studies confirm that the Ni3+ and O- species are at the surface relative to Ni2+ and O2-.The chemical modification caused by prolonged exposure to X-rays on a series of para-substituted phenyl moieties (-NO2, -CN, -CHO, -COOH, -CO2Me, and -(CO2Bu)-Bu-1) at the surface of thiolate-Au self-assembled monolayers (SAMs) has been investigated by X-ray photoelectron spectroscopy (XPS). It is suggested that Ni3+ is formed in a surface redox reaction from Ni2+ with the simultaneous generation of O.

The latter unexpectedly exhibit a high relative proportion of Ni3+ states. The mixed valence states, Ni2+ and Ni3+, are a characteristic feature not only of oxygen interaction with Ni(210) and Ni(100) at low temperature but also of the surface of bulk nickel oxides. Ni (L2, 3 M4, 5 M4, 5) Auger spectra and also recent theoretical work support this conclusion. It is suggested that the initial stage of oxygen chemisorption involves only the Ni(4s4p) band while the Ni(3d) electrons participate in oxide formation, which is reflected by the emergence of Ni2+ and Ni3+ states. There were two distinct regimes of oxygen interaction for low oxygen coverages, θ 0.6 the Ni(2p) difference spectra indicated that both states were formed, their relative proportions being dependent on temperature and coverage. These values were established by the generation of difference spectra in the Ni(2p) spectral region and assignments confirmed by studies of bulk nickel oxides of different stoichiometry. The binding energies of the latter are 854.7 and 856 eV respectively.

O(1s) spectra distinguished between the formation of O- and O2- species while Ni(2p) spectra revealed the formation of Ni2+ and Ni3+ states. The chemisorption of oxygen at Ni(100) and Ni(210) surfaces has been studied by X-ray photoelectron spectroscopy in the temperature range 77-300 K.